How to check if your product is infringing on any IP right?
New technological products are being launched at such high speed that you cannot keep track of the latest entrants. It’s inspiring, to say the least. But shouldn’t companies pause before they take that leap of introducing a new product. Why? Because the commercialisation of these products may be obstructed due to the intellectual property rights of competitors in the industry. The smart thing to do is to perform a Freedom to Operate search at the conceptualisation stage itself. This will let you know if you are infringing on any IP rights directly or indirectly.

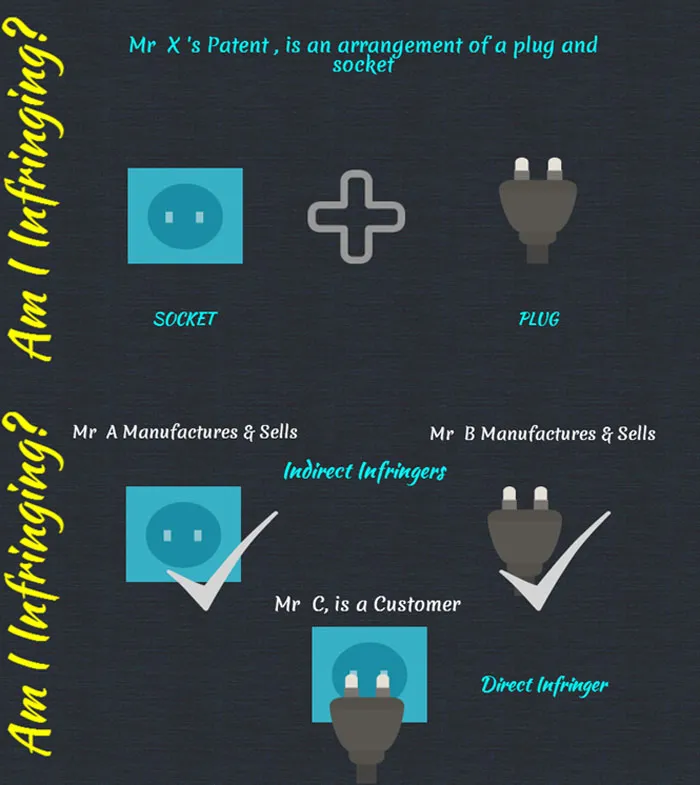
Many times technocrats assume that if they are not infringing on any IP directly, then they are not considered as infringers. However, this is not the right assumption. Let us consider a scenario where a third-party holds a right to an arrangement of a plug and socket. The following questions can be raised:
- If a single company manufactures the plug component separately and the socket component separately and sells them separately, would the company be considered as an infringer? Yes, they will be considered as an infringer even though they are manufacturing them separately and selling them separately, as the intention is not genuine here. The company is just making a sham to manufacture and sell them separately.
- If there are two different manufacturers, who are unrelated; and one company manufactures the plug component and the other company manufactures the socket component, would both the companies be considered as infringers? Yes, both the manufacturers would be considered as infringers; they would be considered as indirect infringers but not direct infringers. In this case, the direct infringer would be the end users who will use the arrangement of the plug and socket. However, the two manufacturers are still considered as infringers as they are enabling the end user to infringe on the plug and socket arrangement by providing the plug and the socket.
The degrees of freedom required to be checked before moving forward are:
- Are you free to manufacture a product? Or, are you free to manufacture a product by a particular process?
Manufacturing is a process which involves the use of various ingredients and a particular process, to manufacture a final product or an intermediary product. Various questions can be raised here:
- Are we free to use a particular ingredient for manufacturing a particular product?
- Are we free to use a particular ingredient in a particular manufacturing process?
- Are we free to manufacture a particular product through a particular manufacturing process?
For example, a pharmaceutical product is manufactured using a specific process that is patented, so as a third person, one begins to manufacture a similar product with a slightly different process. In this case one would not be infringing the process patent, but if specific ingredients of the pharmaceutical product are patented, and the different process involves the use of one of the patented ingredients, then it would be considered an infringement.
- Are you free to use a product or process? Or, are you free to use a product produced using a particular process?
“Using a product or process” can be:
- The use of an end product like in the case of washing machines, refrigerators, etc.
- A particular way of servicing an end product
- The use of an intermediary to manufacture a particular product
- The use of particular manufacturing process
- A particular way of using a product or a system, like a way a gadget or a system of gadgets has to be played
- Are you free to market a product or service? Or, are you free to market a product produced using a particular process?
“Marketing is more than offering for sale”. Even, if someone promotes his product or services by supplying a benefit, it can be an induced infringement. With respect to the same patent example mentioned above regarding patent protection for a specific set of ingredients, if a third-party advertises their product and mentions one of the patented ingredients as a powerful ingredient in their product, or they market a patented ingredient that has simplified the process of manufacturing a similar pharmaceutical product, they are considered as infringers.
- Are you free to sell a product or service? Or, are you free to sell a product produced using a particular process?
“Selling” is a function which happens through different channels involving distributors, retailers, and agents. The product may be an unfinished good, a good manufactured through a particular process, a finished good, a good along with servicing warranty, etc. Anyone or everyone in the sales channel may be responsible for patent infringement. Hence, a degree of freedom is required against each entity in the sales channel.
- Are you free to export a product or service? Or, are you free to export a product produced using a particular process?
If you are not free to “sell”, it is highly unlikely that one can export it out of the country. But then again, can you export an unfinished product, or may be an unassembled one? It is a very subjective question, which can only be determined after examining the third-party patents under question.
- Are you free to import a product or service? Or, are you free to import a product produced using a particular process?
As for import, the same reasoning as export applies. If you are not allowed to sell or export, it is highly unlikely that you would be allowed to import. However, it needs to be identified as to which parts of a product you are prohibited from importing; an intermediate product, or the whole product itself.
Each of these degrees of freedom has to be judged with specific user cases to determine absolutely whether infringement is probable or not. These opinions are best given by experienced professionals who understand the scenario presented to them. Start-ups have great enthusiasm and zest towards their ventures. Therefore taking an extra step of opting for an expert opinion regarding Freedom to Operate may come in as a boon, as opposed to facing unexpected infringement suits in the future.
About the authors:
This post is co-authored by Gaurav Singhal and Ananya Dhuddu. Gaurav is the Director and Principal IP Attorney at Patracode Services Pvt Ltd. A B.Tech in Computer Science, LLB from IIT-Kharagpur and Masters in Business Laws from National Laws School of India University, Gaurav has been working in the IPR field since many years. Ananya Dhuddu is an IP Analyst at Patracode Services Pvt. Ltd. She is a Life Sciences graduate from Pennsylvania State University, USA. She holds a PG Diploma in Medical Law and Ethics from National Law School of India University, Bangalore and a PG Diploma in IPR and Patent Management from GIIP, Bangalore.
(image credit: Shutter Stock)











